 Or you can just remember that the transition metals are represented as groups 3 to 12. So just remember you could do with long way where you give the electron configurations of each transition metal and look at the number of total s and the electrons. 
OK, so just remember, if you're looking at the full out periodic table that I talked about in earlier chapters, you know that the transition metals are also represented as groups 3 to 12 and that new group number designation is also equal to the number of valence electrons. Group Three A is group 13 all the way to group 18 for Group eight A. Group won a Group one group, Two Ways group, too. But they also have another set of numbers that describe them. Remember our main group elements groups one a to eight a. This would be Group three Group 10, 11 and 12. 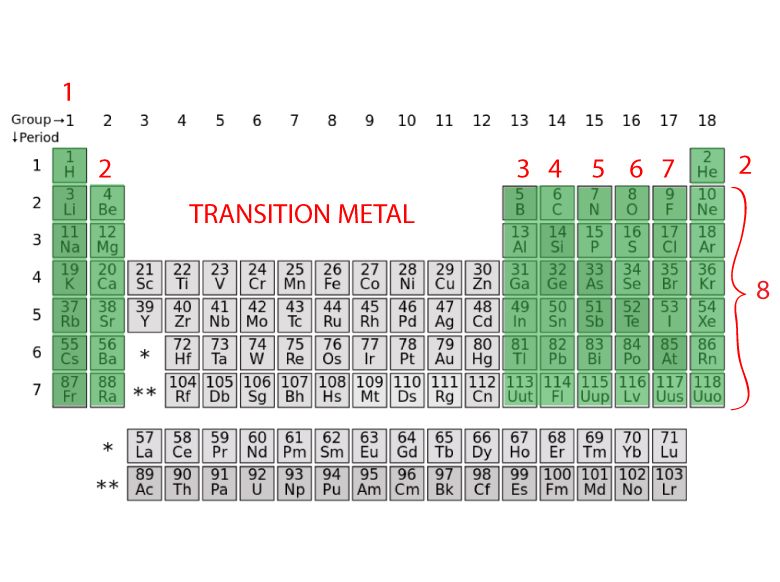
If you look at a full, out electric periodic table that we did in later in earlier chapters, you'll see that these groups also have another set of numbers for their group identity. Remember, besides classified transition metals is Group B elements. And then when we get to zinc, we see that it is two plus 10, which is 12. If we look at them, this is two plus one, which is three, two plus two, which is four, and it goes on and on. I've listed the outermost s orbital's and a deep orbital electrons for each of the transition metals. And here remember your transition Metal elements are known as your group B elements, so we know there's three b all the way to one Be all the way to eight b, but you don't look at that to determine the number of valence electrons. Now with transition metals were going to say the number of Valence electrons equals their outermost s orbital electrons, plus their deep orbital electrons. And when you look at the visual representation of the chlorine atom, you'd also see that there are 10 inner core electrons Hide transcripts Because when you add up these numbers, it gives you 10. If you subtract that, then what's left will be your inner core electrons. But for simplicity, just say group number of the Element main group element is 78 so it has seven valence electrons. So here we have the visual representation of the atom or using the electron configuration to justify this representation of the chlorine atom. So we have one, 2345 six, and then seven. So let's add the additional two electrons that we need. And then we can say here that the outer shell has seven electrons total in it. The second shell, and equals two has a total of eight electrons in it. If we have a representation of the atom like we do here, the first shell has two electrons in it. So here this will be shell one shell to and shell three. Remember, the number in front tells us the shell number. It's electron configuration is one s to to us to two p 63 s 23 p five. And we're going to say here that your atomic number can be thought of as your total number of electrons and that equals just all your valence electrons, plus your inner core electrons. The inner core electrons are just all the remaining electrons that are not valence electrons. 
When we say valence electrons, the valence electrons are just your outer shell electrons involved in forming chemical bonds. So basically not the transition metals were going to say the number of Valence electrons equals the group number of the element. Now when it comes to Valence, electrons were going to say that for main group elements. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
